
GMOs, seeds, pesticides, transparency… European law under attack!
In less than two weeks, between 10 and 19 December, 2025 ended with a rather unprecedented offensive by the European Union authorities against several European texts protecting the environment, health and citizens’ rights. GMOs, pesticides, seeds, corporate responsibility… everything is being called into question in order to “simplify” life for businesses in Europe. Faced with this frenetic pace, Inf’OGM continues its watchdog work, which is essential for public debate, the proper functioning of democracy and the protection of life, by providing accurate, rigorous and reliable information.

The Council of the EU wants to maintain the patentability of GMOs/NGTs
On 19 December 2025, in a still provisional text, the Council of the European Union maintained the patentability of GMOs derived from new genetic techniques (NGT). To this end, it relied on existing law and called for voluntary, but non-binding, commitments, without taking into account the impact on farmers and small seed producers. This text still needs to be validated, amended or rejected by the European Parliament, which had voted against these patents in 2024 and will have to take a position in 2026 without being able to propose new amendments.

Agricultural biodiversity at risk with new seed regulation
On 10 December 2025, the Council of the European Union approved a mandate to begin negotiations with the European Parliament on a new seed regulation. Many civil society organisations have criticised the draft regulation for failing to protect small and medium-sized breeders, seed producers and farmers. They are calling for a diverse seed market, the implementation of farmers’ rights to seeds and guaranteed access to varieties suited to their farming systems.

New GMOs to produce medicines tested in Spain
Near Valencia (Spain), in Polinyà del Xúquer, a trial of genetically modified tobacco to produce a molecule of therapeutic interest has been authorised. This tobacco has been modified using genetic modification techniques presented as new (NGT). The two partners in charge of the trial, the company MadeInPlant and the agricultural union AVA, declared the trial in December 2024, in accordance with Directive 2001/18 governing GMOs, even though they claim that this tobacco is not a GMO but an NTG plant.

Qualified majority in the Council of the European Union to deregulate numerous GMOs
On Friday 19 December, the Member States meeting within the Council of the European Union reached a qualified majority agreement on a text to deregulate many GMOs. This text, negotiated two weeks earlier between representatives of the European Commission, the European Parliament and the Council, proposing broad deregulation of GMOs obtained through new techniques of genetic modification, has finally convinced a sufficient majority of States. The European Parliament is now due to consider it in January 2026.

The majority of micro-organisms modified by NGTs are detectable
In 2025, the European network of GMO laboratories (ENGL) published a report written by several of its experts on the detection and identification of genetically modified microorganisms using new techniques in the food and feed industry. According to this report, the vast majority of these micro-organisms are detectable and identifiable. However, “in somecases”, with the processes currently available, very small genetic modifications can be more complicated, or even impossible, to detect and differentiate from those that can occur without technical intervention on the genome.
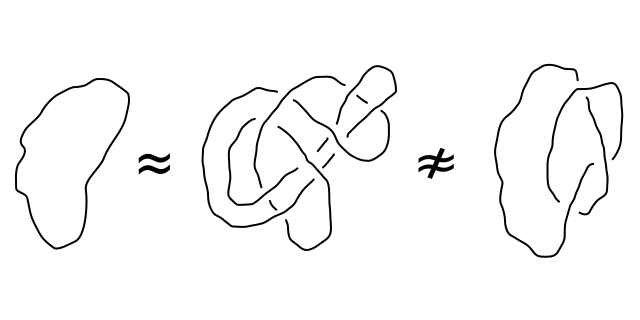
GMO/non-GMO equivalence: the Commission turns “certain cases” into a general rule
The proposal to deregulate some of the GMO plants made by the European Commission in July 2023 is based in particular on the assumption that new techniques of genetic modification can produce organisms with modifications that could also be obtained using so-called “conventional” methods. To make this claim, the European Commission uses a subtle but decisive semantic abuse in its proposal…

NGT regulations: trilogue of the deaf under pressure from Denmark
Negotiations on the future European regulation on new genetic modification techniques have been focusing on two sensitive issues for several months: patentability and sustainability. Keen to conclude the dossier before the end of the year, the Danish Presidency is stepping up efforts to find a compromise, at the risk of neglecting issues that are of particular concern to small and medium-sized breeders and farmers. Denmark will seek an agreement this week, having already threatened to freeze discussions and refer the text back to the European Parliament for a second reading.
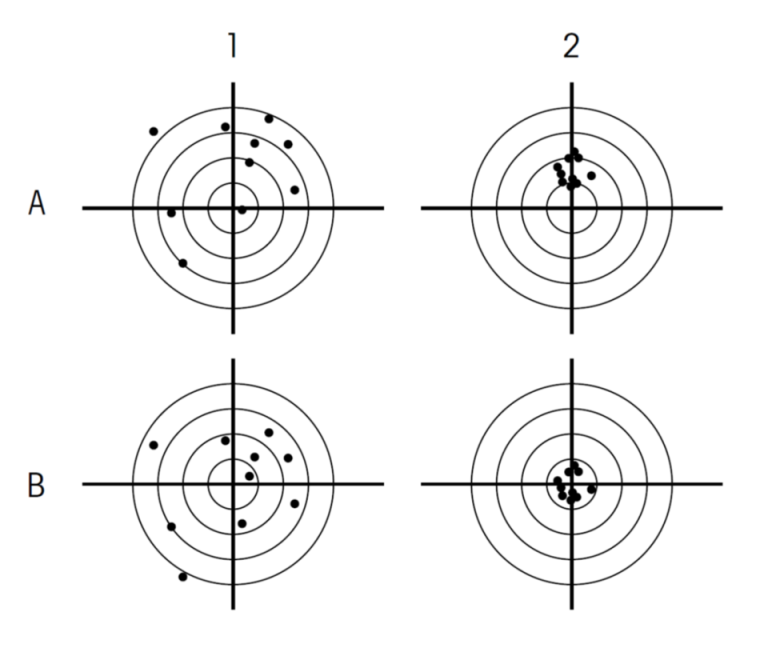
Directed, targeted, precise mutagenesis… Are these adjectives misleading?
Being precise, targeting and achieving one’s goal, directing a mutagenesis… these are adjectives that convey a sense of control and precision. However, on closer inspection, these adjectives mean nothing in a legal text. Because, in the European Commission’s proposal to deregulate a number of GMOs, they are not accompanied by their corollaries: targeted where? Precise to what degree? Directed by what or by whom?

Consumer associations call for continued labelling and traceability of GMOs
On 14 October 2025, eight consumer associations from various EU Member States published an opinion calling on European institutions to maintain GMO labelling to enable European consumers to make informed choices about their food.

GMO/NGT Regulation: civil society organisations concerned about the outcome of the trilogue
As the trilogue on the regulation of new genomic techniques (NGT) continues, civil society organisations are expressing their concerns about the outcome of the discussions, particularly on the issue of patents. This is evidenced by two recent position statements: those of the European Coordination Via Campesina (ECVC) and Arche Noah, which illustrate their mobilisation around this crucial debate at a key moment when European decisions are being made.

The European Commission is more attentive to biotech companies than to citizens
In early August, the European Commission launched a multilingual online public consultation on its forthcoming “Biotechnology Regulation”. Presented as an exercise in transparency and citizen participation, the questionnaire is in fact primarily designed to gather the industry’s position. In particular, it does not address the ethical and civic dimensions raised by new genome editing techniques (NGTs), such as health risks, the appropriation of living organisms by industry, or the management of health data. This regulation could offer the Commission and multinationals a way out if current or past negotiations on other legislative acts fail to achieve their objectives.

